 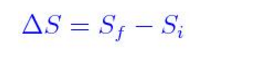 
If temperature changes during the process, then it is usually a good approximation (for small changes in. In 1865 Rudolf Clausius expanded his previous work from 1854 on the concept of "unkompensierte Verwandlungen" (uncompensated transformations), which, in our modern nomenclature, would be called the entropy production. The SI unit for entropy is joules per kelvin (J/K). The importance of avoiding irreversible processes (hence reducing the entropy production) was recognized as early as 1824 by Carnot. The knowledge of the absolute entropies of substances allows us to calculate the entropy change (So) for a reaction. Any chemical or physical change in a system may be accompanied by either an increase in entropy (S > 0) or a decrease in entropy (S. Standard entropies for a few substances are shown in the table below. Changes in entropy (S), together with changes in enthalpy (H), enable us to predict in which direction a chemical or physical change will occur spontaneously. Development of entropy in a thermodynamic systemĮntropy production (or generation) is the amount of entropy which is produced during heat process to evaluate the efficiency of the process.Įntropy is produced in irreversible processes. Entropy and the Second Law of Thermodynamics. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
